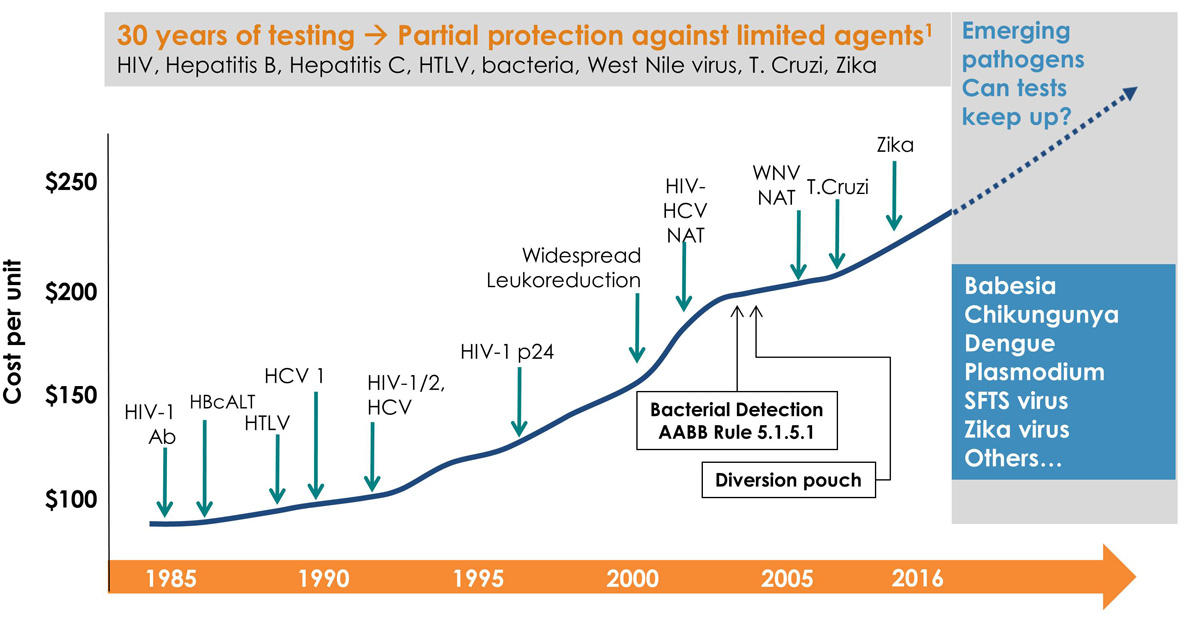
Infectious Risk
Choose Transfusion-Ready, Pathogen-Reduced Platelets. Today.Every day, blood transfusions save thousands of lives around the world.
These transfusions, however, are not without risk.1
Blood can contain potentially harmful elements which can be transmitted to the patient, such as bacteria, viruses (Zika, West Nile Virus, Chikungunya), parasites, and emerging pathogens. Thorough testing can reduce some of the known infectious risks, but these tests do not always detect emerging or re-emerging diseases.
Bacteria
~1:1500-2500 platelet units are estimated to contain infectious bacteria despite contamination testing.1,2
- Transfused patients receive ~6 platelet units, as is common in oncology care, increasing the potential risk of receiving a contaminated unit to ~1:250-4002,3
Conventional platelets are susceptible to bacterial contamination, room temperature storage facilitates bacterial growth.
Read the FDA Guidance for Bacterial Mitigation.
- Sensitivity of primary culture is only 26-44%1,4,5
- Utility of secondary testing is constrained by its limit of detection6
Sepsis and bacterial infections attributed to platelets are underrepresented due to passive reporting2
- Passive surveillance for septic transfusion reactions rarely identifies events2 (case study example link to page below)
- Active surveillance at a major academic institution for bacterially contaminated platelets showed that patients continued to receive contaminated platelets, had septic transfusions reactions and yet, all were missed.2
- Hospital-acquired infections are associated with platelet transfusions7
- Colonized central lines can be caused by contaminated platelets8
- Reduce your patient’s risk of transfusion-associated sepsis with INTERCEPT® Blood System treated platelets
Viruses
In the age of global travel, and global warming propagating the vectors for disease, the impact of a sudden viral outbreak and necessary quarantine leading to the destabilization of the blood supply is a real concern. Unlike bacteria, where culture can pick up identified and unidentified bacteria (depending on sample and aerobic or anaerobic growing conditions), nucleic acid testing (NAT) for viral detection is specific to an identified virus. Identification and NAT development takes time.
The emergence of Zika virus, for example, is thought to cost US blood centers $100 million annually1. A global analysis of the substantial differences in costs and health benefits of serology and NAT screening of donations for blood transfusion in different Western countries for HIV, HCV and HBV show that even already developed tests have high cost burden2. Not only does a reactive approach such as serology and NAT testing require time to develop, the incremental costs associated with each new virus substantially impact the availability and sustainability of blood products.
INTERCEPT has been called upon to help sustain platelet availability during arboviral outbreaks, such as chikungunya, West Nile and most recently, Zika viruses.
- 2005 CHIKV epidemic on Ile de La Réunion, France3
- CHIKV outbreaks in Guadeloupe and Martinique, French Polynesia4,5
- 2014 CHIKV and DENV outbreaks in the Caribbean region6
- 2016 WHO,7 US FDA,8 ECDC9 issued guidances recommending pathogen reduction as an option to mitigate risks related to ZIKV outbreaks.
Zika Virus
Only 20% of Zika virus-infected individuals are symptomatic, therefore, the potential for transfusion transmission from an asymptomatic blood donor is high.10 Cases of transfusion-transmitted Zika were documented during the 2015-2016 outbreak in Brazil11 despite efforts to screen donors and test components.
Estimated potential range of Aedes aegypti in the United States, 20179
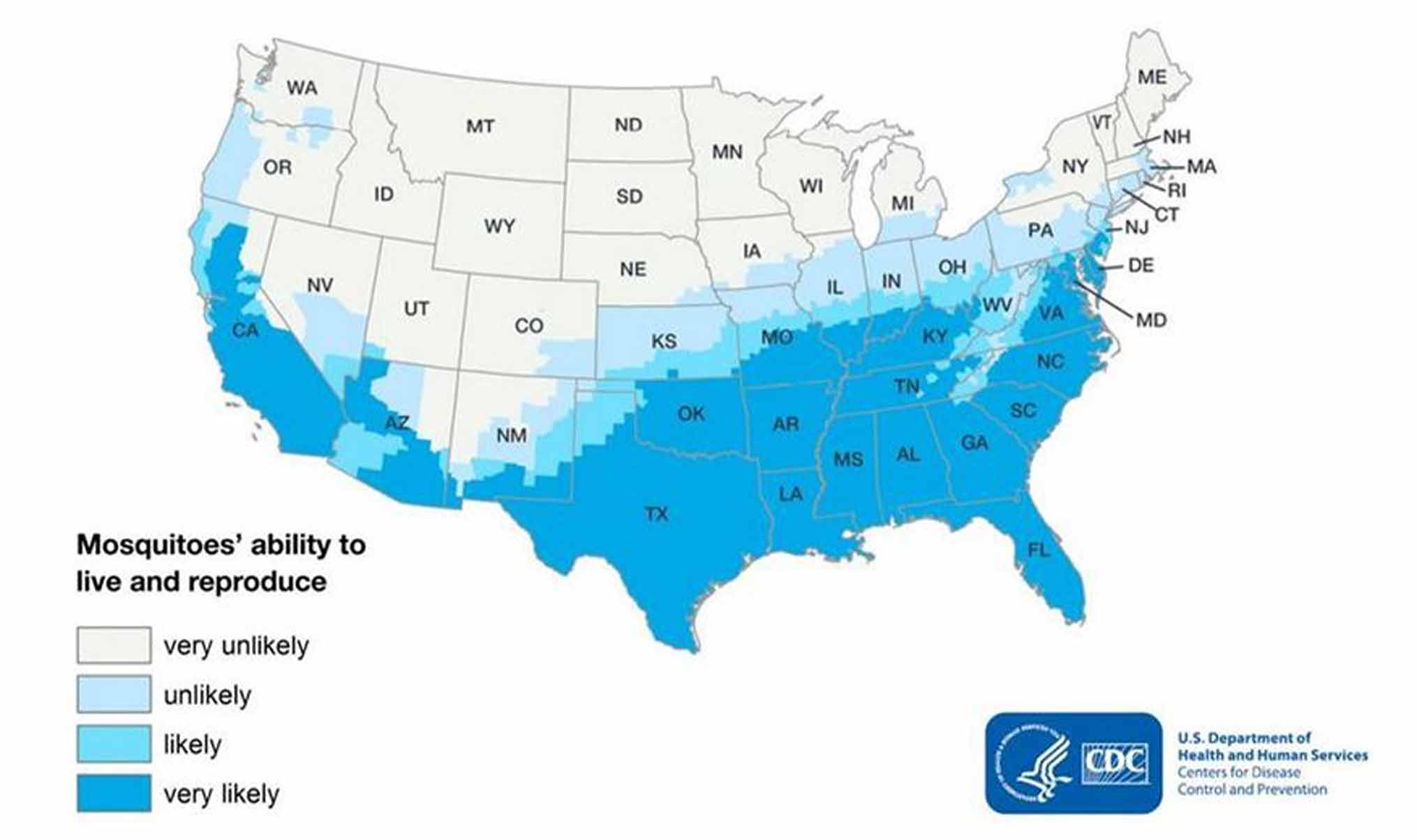
Visit the CDC site to view this map.
Industry Guidances Issued
Pathogen reduction may be implemented rather than testing donations: FDA Recommendations for Reducing the Risk of Zika Virus Transmission by Blood and Blood Components – Guidance July 2018 (Guidance withdrawn as no longer relevant)
Pathogen reduction technology (PRT) may be implemented: World Health Organization (WHO) – Maintaining a safe and adequate blood supply during Zika virus outbreaks – Interim Guidance February 2016
*It should be noted, Zika NAT sensitivity and specificity fails frequently in minipool testing, ensuring safety requires every donation be tested individually, a time and cost-intensive process.12
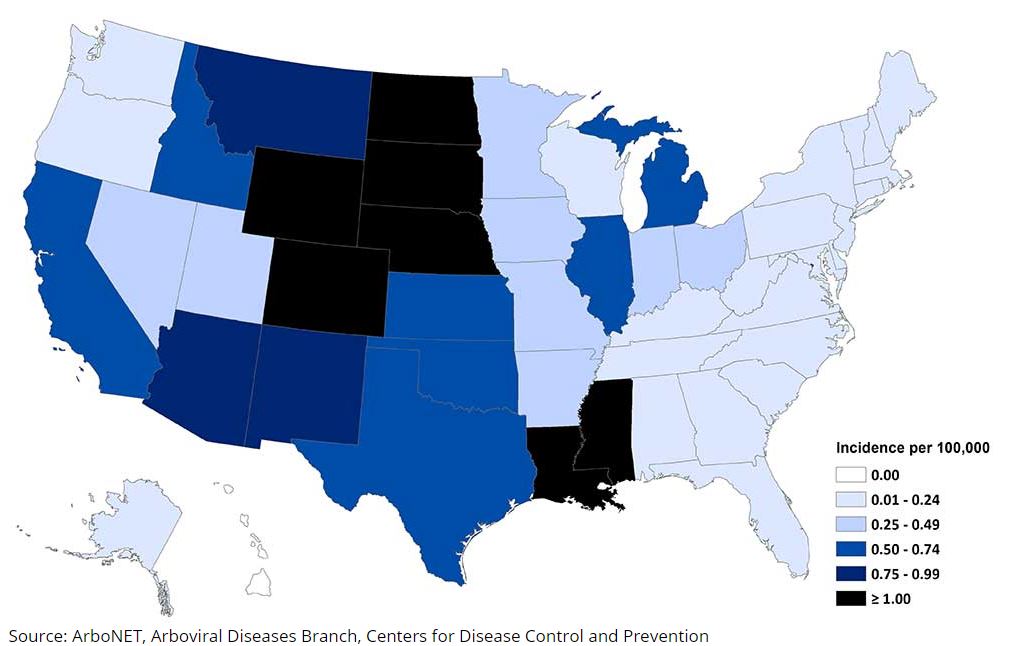
West Nile Virus
Three years elapsed between emergence of West Nile Virus (WNV), identification of it being a transfusion-transmitted infection (TTI) and the introduction of a WNV specific NAT. In this time, several hundred transfusion recipients are estimated to have been infected.13
Once the WNV NAT was developed (2003), testing of minipools of blood donations prevented hundreds of cases of WNV infection and led to the prospective identification of 519 donors who were positive for WNV RNA, removing more than 1000 potentially infectious related components from the blood supply of the American Red Cross.14
Unfortunately, WNV NAT fails to detect units with a low level of viremia, some of which were antibody-negative and infectious15. Although data supports the use of targeted nucleic acid amplification testing of individual donations in high-prevalence regions13, this practice is currently only used seasonally and after a triggering event.
Average annual incidence of West Nile virus neuroinvasive disease reported to CDC by state, 2019
Chikungunya
During the 2005-2007 chikungunya virus (CHIKV) epidemic on Ile de La Réunion, France, more than 30% of 750,000 inhabitants were infected. Local blood donation was suspended to prevent transfusion-transmitted infection (TT-CHIKV) 16. To sustain the availability of platelet (PLT) components, the Établissement Français du Sang implemented universal pathogen inactivation (INTERCEPT Blood System, Cerus Europe BV) of PLT components (CPAs).3 While local infection has not been documented in the US, almost 250 travel-associated cases were reported in 2016 and the mosquito by which chikungunya is spread from human to human, Aedes aegypti and Aedes albopictus, can be found in the southern and coastal states.17
A follow-up study assessed the safety of INTERCEPT-treated platelet units transfused in routine clinical practice to neonate, pediatric and adult patients during the Ile de La Réunion chikungunya outbreak. There were no cases of TT-CHIKV, no serious adverse events, no cases of transfusion-transmitted sepsis and no deaths due to INTERCEPT platelet transfusions.3
Parasites
- Remain stable in the platelet unit
- Retain infectious risk
- Metabolize nutrients, potentially altering pH
Causes of Parasitic Infections
Recipients of platelets may be infected by donor-derived parasites in the platelet concentrates (the donors may be asymptomatic during donation). Transfusion-transmitted infections of parasites replicating in red blood cells (Babesia and Plasmodium species) as well as parasites residing in the bloodstream (Trypanosoma cruzi) have been reported.
Impact to Patients
Infection with Babesia and Plasmodium species causes malaria symptoms such as haemolytic anaemia, thrombocytopenia, fever and chills, which can affect the health of transfusion recipients and lead to fatalities. Infection with Trypanosoma cruzi causes Chagas’ disease, which often develops into a severe acute phase infection in immunocompromised patients (of which many receive transfusions) causing fever, myocarditis, and/or meningoencephalitis with a lethality of 10%.
Industry Guidance Issued
Pathogen reduction may be implemented as an alternative to testing or Babesia-related donor screening questions, as described in FDA Recommendations for Reducing the Risk of Transfusion-Transmitted Babesiosis. The FDA also includes pathogen reduction in its recommendations to reduce the risk of transfusion-transmitted malaria, as an alternative to certain travel-related deferrals.
Be proactive, not reactive against emerging pathogens with INTERCEPT
Emerging Pathogens: Is a Reactive Approach Enough?
Conventional Testing:
- Not proactive, tests are subject to lengthy lead times and delays associated with development and regulatory approval1,2
- Viruses are spreading with population travel, migration and climate changes1,3,4
- Mosquitos and tick-borne viruses are on the rise5,6
- Availability of commercially-approved screening tests may be limited or non-existent4,7
- The first tick-borne parasite to have an FDA approved blood test was babesia, in 20188
The Global Virome Project predicts there are (currently) 1.3 million unidentified viruses and that some 500,000 are likely to be zoonotic: able to jump from other animals to humans9. In five years, they have discovered 820 novel viruses. It’s hard to predict what unknown viruses are already common in humans and in our companion animals or what diseases they might cause in the future9. As these viruses evolve and mutate, the time, difficulty, and costs, associated with detection test development threaten the stability of the blood supply.
Bacteria References
1. Dumont, L.J., et al., Screening of single-donor apheresis platelets for bacterial contamination: the PASSPORT study results. Transfusion, 2010. 50: p. 589-599. 2. Hong, H., et al., Detection of septic transfusion reactions to platelet transfusions by active and passive surveillance. Blood, 2016. 127(4): p. 496-502. 3. Kleinman, S., W. Reed, and A. Stassinopoulos, A patient-oriented risk-benefit analysis of pathogen-inactivated blood components: application to apheresis platelets in the United States. Transfusion, 2013. 53(7): p. 1603-18. 4. Murphy, W.G., et al., Screening platelet concentrates for bacterial contamination: low numbers of bacteria and slow growth in contaminated units mandate an alternative approach to product safety. Vox Sang, 2008. 95(1): p. 13-19. 5. Pearce, S., G.P. Rowe, and S.P. Field, Screening of platelets for bacterial contamination at the Welsh Blood Service. Transfus Med, 2011. 21(1): p. 25-32. 6. Platelet PGD® Test [Package Insert]. April, 2017, Verax Biomedical: Marlborough, MA. 7. Aubron, C., et al., Is platelet transfusion associated with hospital-acquired infections in critically ill patients? Critical Care 2017. 21(1): p. 2. 8. Yousif, A., M. Jamal, and I. Raad, Biofilm-based central line-associated bloodstream infections. Adv Exp Med Biol, 2015. 830: p. 157-79.
Virus (including Zika Virus, West Nile Virus, and Chikungunya) References
1. Ellingson, K., et al., Cost projections for implementation of safety interventions to prevent transfusion-transmitted Zika virus infection in the United States. Transfusion, 2017. 57(S2): p. 1625-1633. 2. Janssen, M., et al., An assessment of differences in costs and health benefits of serology and NAT screening of donations for blood transfusion in different Western countries. Vox Sang, 2017. 112(6): p. 518-252. 3. Rasongles, P., et al., Transfusion of platelet components prepared with photochemical pathogen inactivation treatment during a Chikungunya Virus epidemic in Ile de La Reunion. Transfusion, 2009. 49: p. 1083-91. 4. Aubry, M., et al., Chikungunya outbreak, French Polynesia, 2014. Emerg Infect Dis, 2015. 21(4): p. 724-6. 5. Dorleans, F., et al., Outbreak of Chikungunya in the French Caribbean Islands of Martinique and Guadeloupe: Findings from a Hospital-Based Surveillance System (2013-2015). Am J Trop Med Hyg, 2018. 98(6): p. 1819-1825. 6. Rico, S., et al., Treatment use study of platelet components treated with amotosalen and ultraviolet A light in response to the chikungunya and dengue epidemic in Puerto Rico – true study interim results. Blood Transfusion, 2016. 14(Supplement 1): p. s 73. 7. Maintaining a safe and adequate blood supply during Zika virus outbreaks. Interim guidance. 2016, WHO. 8. Food and Drug Administration Center for Biologics Evaluation and Research, Revised Recommendations for Reducing the Risk of Zika Virus Transmission by Blood and Blood Components, Department of Health and Human Services, Editor. 2016, US FDA. (Withdrawn May 13, 2021) 9. European Centre for Disease Prevention and Control, Zika virus and safety of substances of human origin – A guide for preparedness activities in Europe, ECDC, Editor. 2016: Stockholm. 10. Goodnough, L. and M. Marques, Zika Virus and Patient Blood Management. Anesth Analg, 2017. 124(1): p. 282-289. 11. Motta, I.J., et al., Evidence for Transmission of Zika Virus by Platelet Transfusion. N Engl J Med, 2016. 375(11): p. 1101-3. 12. Galel, S., et al., First Zika-positive donations in the continental United States. Transfusion, 2017. 57(3pt2): p. 762-769. 13. Petersen, L.R. and J.S. Epstein, Problem solved? West Nile virus and transfusion safety. N Engl J Med, 2005. 353(5): p. 516-7. 14. Stramer, S.L., et al., West Nile virus among blood donors in the United States, 2003 and 2004. N Engl J Med, 2005. 353: p. 451-459. 15. Busch, M., et al., Screening the blood supply for West Nile virus RNA by nucleic acid amplification testing. New Engl J Med, 2005. 353(5): p. 460-7. 16. Brouard, C., et al., Estimated risk of Chikungunya viremic blood donation during an epidemic on Reunion Island in the Indian Ocean, 2005 to 2007. Transfusion, 2008. 48: p. 1333-1341. 17. CDC. Chikungunya Virus: 2016 provisional data for the United States. 2017 [cited 2017 Oct 6]; Available from: https://www.cdc.gov/chikungunya/geo/united-states-2016.html.
Emerging Pathogen References
1. Howard, C. et al. Emerging Microbes and Infections, 2012. 1(e46). 2. Galel, S., et al. Transfusion, 2017. 57(3pt2): p. 762-769. 3. Klein, H.G., et al. Transfusion, 2007. 47: p. 2338-2347. 4. Diuk-Wasser, M. Scientific American, A Division of Nature America. 2013. 5. Morin, C. et al. PNAS, 2013. 110(39): p. 15620-5. 6. Krause, P., et al. Am J Trop Med Hyg, 2003. 68(4): p. 421-436. 7. Yuan, L., et al., A single mutation in the prM protein of Zika virus contributes to fetal microcephaly. Science, 2017. 8. FDA approves first tests to screen for tickborne parasite in whole blood and plasma to protect the U.S. blood supply: New donor screening tests to help reduce risk of transfusion-transmitted babesiosis, CBER, Editor. 2018. 9. Branswell, H. Finding the world’s unknown viruses — before they find us. 2016